You might not think much about what powers your phone or car until the electric bill jumps or a heatwave strains the grid. Behind the scenes, scientists are racing to reinvent how we store electricity, and China has just quietly crossed a new frontier.
A team there has built the first working hydrogen-based solid-state battery, turning a long-discussed idea into real hardware.
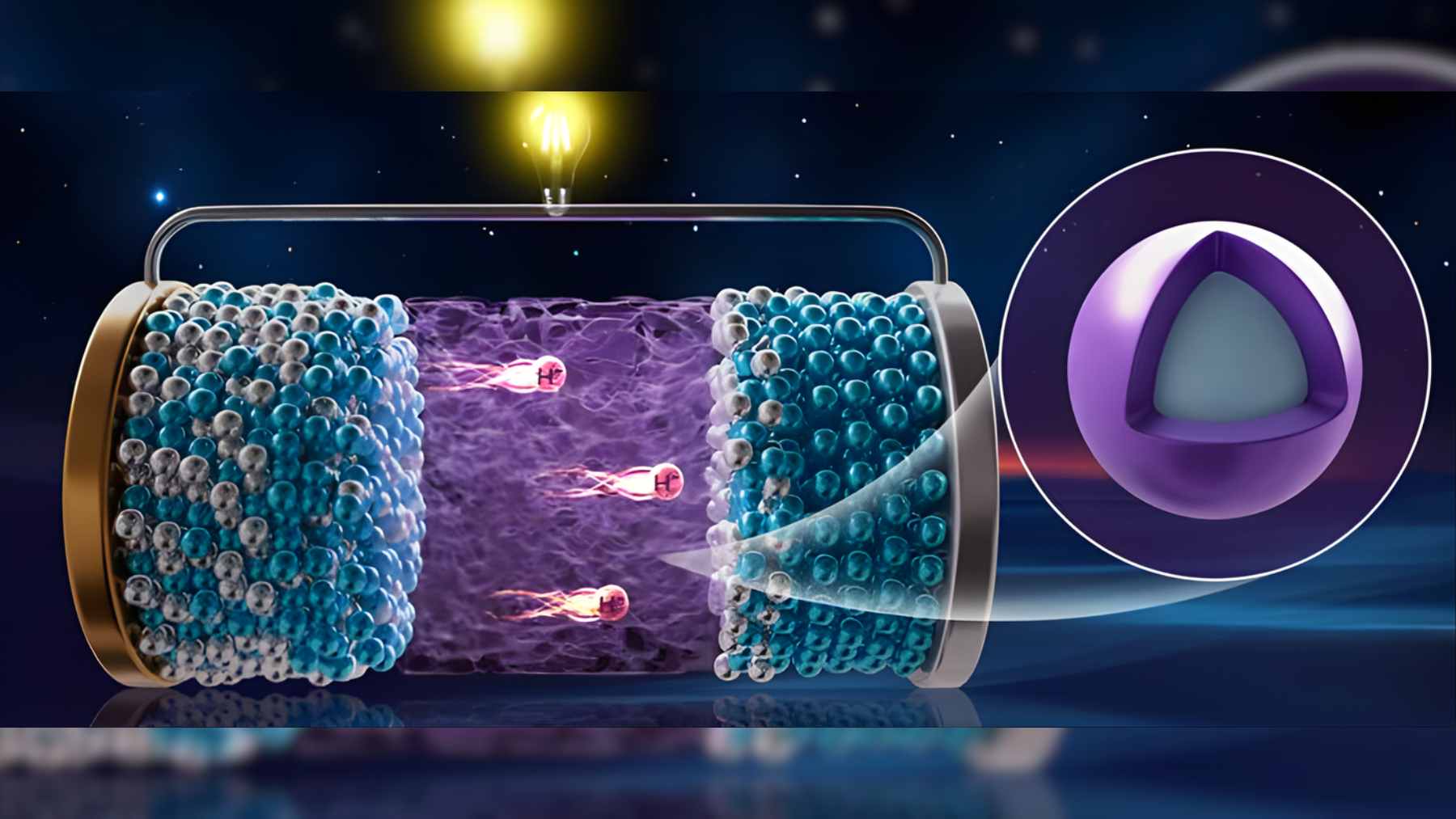
The prototype, described in the journal Nature by researchers at the Dalian Institute of Chemical Physics, part of the Chinese Academy of Sciences, uses hydride ions instead of lithium to carry charge through a solid material.
For a field focused on safety, cost, and raw material supply, that shift could eventually change how we power electric vehicles and store solar or wind energy, even though the technology is still at a very early stage.
What Makes a Hydrogen Battery Special
Most batteries today move lithium ions between two electrodes through a liquid or gel electrolyte. In the new device, the charge is carried by hydride ions, which are hydrogen atoms with an extra electron and often described as H minus. This different carrier alters the chemistry and could enable batteries to store more energy in a lighter package going forward.
The Chinese team has also kept everything in a solid state, so both the electrodes and the electrolyte are solids instead of liquids. Solid-state designs are attractive because they aim to cut leakage and fire risks while packing more energy into the same volume. Moreover, the use of hydrogen in this context helps prevent tiny metal filaments called dendrites from growing and shorting the cell, a critical safety concern in many lithium systems.
Inside the First Chinese Hydride Ion Prototype
The prototype developed by Ping Chen’s group features a positive electrode made from sodium and aluminum hydride, a well-known hydrogen storage material. The negative electrode consists of a cerium and hydrogen compound. Between them is a specially-engineered core-shell composite that combines a cerium hydride core with a thin barium hydride layer to efficiently guide hydride ions.
This core-shell material functions as a solid electrolyte capable of moving hydride ions at room temperature, addressing a significant roadblock in the field. The study indicates that it behaves as an exceptionally good ion conductor above about 60 degrees Celsius, while maintaining chemical stability against the surrounding materials. The researchers noted, “using hydrogen as the charge carrier avoids the formation of dendrites and opens new paths for clean energy storage and conversion.”
Early Performance Numbers and a Simple LED Test
In initial tests, the positive electrode delivered a specific discharge capacity close to 984 milliampere hours per gram at room temperature. After 20 charge and discharge cycles, the cell still provided around 402 milliampere hours per gram, indicating a drop in performance but not a complete collapse. For many experts, these figures signal a promising proof of concept rather than a ready-to-ship product.
In a stacked setup, the operating voltage reached about 1.9 volts. While this is not enough compared to the packs used in electric vehicles, it is sufficient to power a simple yellow LED lamp in the lab. Watching that tiny light turn on is a straightforward moment, but it confirms that the chemistry works beyond computer simulations.
From Lab Bench to the Energy Transition
What will it take to move from a shining lab demo to something that truly impacts your monthly bill? As of now, this hydrogen battery represents a scientific milestone rather than a commercial product.
The cell has only cycled a few dozen times, and engineers still need to demonstrate that it can endure hundreds or thousands of cycles, scale up the materials, manage heat in larger packs, and connect many small cells in series to achieve higher voltages.
Nonetheless, researchers are already envisioning hydride ion batteries as an emerging frontier in energy storage, capable of complementing lithium rather than replacing it overnight. A recent overview in the journal The Innovation points to potential applications ranging from smartphones to electric vehicles and grid storage systems, particularly where safety and material abundance are crucial.
Ultimately, this breakthrough from China illustrates that ideas once relegated to theoretical discussions can now be physically manifested to power tangible devices. If future developments address durability and cost challenges, hydrogen-based batteries could one day contribute to smoothing out solar energy after sunset and reducing electric vehicles’ reliance on scarce metals.
The main study has been published in the journal Nature.